Translate this page into:
Challenges and solutions in the days of COVID-19: Experiences from a tertiary referral centre
*Corresponding author: Neethu Abraham, Department of Dermatology, Venereology and Leprology, T.D. Medical College, Alappuzha, Kerala, India. drneethuabraham64814@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Abraham N, Sobhanakumari K. Challenges and solutions in the days of COVID-19: Experiences from a tertiary referral center. J Skin Sex Transm Dis 2020;2(2):70-4.
Abstract
COVID-19 is not a lung disease alone. It produces several collateral damages in all the specialities of medicine, including dermatology. Neither a major breakthrough in the treatment nor a vaccine has been introduced to deal with this virus. We developed a three-pronged strategy to ensure that our patients are not denied treatment for their chronic dermatoses; at the same time, minimizing their risk of contracting the COVID-19 infection through frequent hospital visits. First, we reduced the need of the patients for repeated hospital visits by providing them medical help through phone calls. Second, we ensured safety of the patients who comes for dermatological procedures, photo therapy, and laser therapies. Third, we ensured the protection of our lab persons by following strict guidelines while performing blood tests of COVID-19 patients.
Keywords
COVID-19
Vesiculobullous
Dermatological procedures
Blood test
Phototherapy
INTRODUCTION
The 21st century is witnessing one of the biggest challenges faced by humankind. COVID-19 has become one of the most dangerous viruses, to date affecting almost all the organs including skin.[1] In this “corona era,” should this be the only matter of concern for a dermatologist?
We are facing several inevitable collateral damages in our day-to-day dermatology practice, as a part of the COVID-19 outbreak.
COLLATERAL DAMAGES OF COVID-19 IN THE FIELD OF DERMATOLOGY
Inability of the patients to attend the outpatient department (OPD) for their regular follow-up[1]
Difficulties to carry out dermatological procedures including laser treatment and phototherapy
Risks associated with doing blood tests such as Venereal Disease Research Laboratory (VDRL) test in COVID-19 cases. (Unlike the other blood and body fluid investigations which are carried out in the central laboratory of our institution, VDRL test of the whole institution is performed in the laboratory attached to the outpatient clinic of dermatology department)
Dermatological problems among health care workers and COVID-19 patients.
Inability to attend the OPD for follow-up
There are several reasons contributing to this:
Nationwide lockdown and restricted transportation facilities
The “corona phobia” of going to a hospital where the number of COVID-19 patients is on the rise
Financial constraints limiting the hospital visits of patients, as most of them are left jobless during the lockdown.
This may lead to irregular follow-up which can adversely affect the management of chronic diseases that require strict treatment compliance and regular monitoring.
We experienced maximum difficulty in the management of patients with vesiculobullous diseases.
WHY “VESICULOBULLOUS DISEASES?”
Patients with vesiculobullous disease develop exacerbations and become unresponsive to retreatment if they miss the regular therapy. They are usually on steroids alone or in combination with rituximab or other immunesuppresants. After discharge from the hospital, all these patients are advised strict follow-up with regular reviews every 2–4 weeks. Majority of these patients were from the low socioeconomic strata and used public transport to visit the hospital. Travel by public transport measures may put them at a greater risk for the COVID-19 infection considering their immunosuppressive state. Moreover, the follow-up visits became almost impossible with the subsequently announced stringent lockdown measures.
Thus, we started a journey with our patients of vesiculobullous diseases – to help them tide over this crisis without compromising their dermatological quality of life.
Our strategy of managing these patients was as follows:
From the hospital registers, we collected the details of all the patients with a diagnosis of vesiculobullous disease, with special attention to those admitted during the past 1 year (March 15, 2019–March 15, 2020).
There were 38 patients, that is, 21 cases of bullous pemphigoid, 13 cases of pemphigus vulgaris, three cases of pemphigus foliaceus, and one case of paraneoplastic pemphigus. Six patients (four bullous pemphigoid and two pemphigus vulgaris patients) could not be reached over the phone. We sought the help of their respective nearest primary health centers to trace these patients, but could not reach them owing to incorrect or incomplete address or probable migration to another place. All the 32 patients who could be contacted were on systemic steroids. One was receiving methotrexate and three were receiving azathioprine in addition.
We contacted them on March 16, 2020. Most of them shared their apprehensions about COVID-19 outbreak and their fear of a hospital visit. They were reassured and were given the contact number of a resident doctor/faculty for clearing their queries if required.
Over the phone, we enquire about their health status, details of the treatment they were receiving at the time and details regarding their nearest health center to ensure drug availability. The required dose modifications, complications that may arise on abrupt withdrawal of the drugs and the relevant investigations were advised over the phone. All the patients, especially diabetics, were advised to check their random blood sugar once in 2 weeks which they were directed to inform us during the telephonic follow-up we carried out once in 2 weeks. For patients receiving methotrexate or azathioprine, we recommended complete hemogram and liver function tests once a month. Patients were given the option to send the photographs through WhatsApp if new lesions develop.
Since then, every 2 weeks, we are calling them regularly and advising the needful.
From March 16, 2020, to June 16, 2020, we had three more inpatient admissions – two pemphigus vulgaris cases and one case of pemphigus foliaceus. These patients were also included in the list – thus currently 35 patients are under our follow-up.
RESULTS
None of these patients had any severe exacerbations requiring hospital admission during these 3 months.
Three cases of pemphigus vulgaris had relapse, for whom we increased the dose to one step higher than the lowest previous dose at which the disease was controlled.[2] They were enquired about the improvement every 2 days. At present, all the three patients are stable, without any new lesions.
One patient with paraneoplastic pemphigus who was under follow-up for multiple metastatic malignant foci from our radiotherapy department succumbed to death on March 30, 2020, following cardiorespiratory arrest.
We believe this strategy of reaching out to patients may help other patients with chronic diseases such as psoriasis and connective tissue diseases as well.
The next concern was for the other routine outpatients. Under the coordination of district medical office, district legislation, and medical college, a new program was launched on March 28, 2020, named, “The doctor online program.” Patients were informed regarding this program through audiovisual and print media. They could register their names by making a call in the provided phone number. They were advised to give a brief account of their medical complaint during this call. The service was available round the clock. SIM cards were provided for the doctors by the program organizers. A doctor from the health service/medical college would call them first and give the appropriate medical advice.
If the doctor felt a need for expert opinion, the respective specialist on duty was informed, who would call them back and give the necessary advice. This facility was employed in 17 departments including dermatology. The program was a grand success and was well accepted by the public.
Limitation
Telephone and online consultations are helpful during this crisis period but we cannot ignore the fact that nothing can replace the diagnostic accuracy and the warmth of a face-to-face interaction between the doctor and the patient.
Difficulty in doing the dermatology procedures, laser treatment, and phototherapy
We used to do several dermatological and cosmetic procedures, phototherapy, and laser treatment. However, considering the risk of COVID-19, we made the following modifications in our working pattern:
All the cosmetic procedures are withheld completely for the time being. Patients were informed
-
Phototherapy for indications like cutaneous T-cell lymphoma was continued. We follow the following precautions for phototherapy.[3]
Advices: Patient should come alone for the phototherapy. For minor patients, one guardian is allowed to accompany, but social distancing, hand sanitizing, and wearing of masks are mandatory. Every patient should bring their individual hand sanitizer, mask, goggles, and a bag to store their clothes when they disrobe during the phototherapy. After the procedure, this plastic bag should be discarded in the yellow kit provided just outside the phototherapy room. Apply hand sanitizer before entering and after leaving the unit.
Measures to be taken by the Doctor: History taking and clinical examination of the patients to rule out COVID-19 infection. Maintain a time gap of 2 hours between two consecutive phototherapies. To maintain a gap of six feet between the seats in the waiting area. To use hand sanitizer before and after seeing each patient. Avoid turning on the fan in the phototherapy unit. If needed, treatment can be fractionated to avoid excessive heat buildup in the unit. After each patient, disinfect high-touch surfaces of the phototherapy equipment and the changing area with 0.1% hypochlorite. The phototherapy unit should be left open ensuring good ventilation for 2 hours after every patient.
In patients with diagnostic dilemma, skin biopsy becomes mandatory. Despite the risks, we perform the essential skin biopsies after taking a detailed patient history to rule out recent travel/contact with COVID-19 patients/any symptoms suggestive of COVID-19. The biopsies are done (after rapid antigen test of the patient to rule out COVID-19 infection) by the clinician wearing N95mask, face shield, apron, and double gloves.
Risks of doing VDRL test in COVID-19 patients
Another challenge we faced was in performing the VDRL test in COVID-19 patients and suspects. Unlike the other blood and body fluid investigations which are carried out in the central laboratory of our institution, VDRL testing of whole institution was carried out in the laboratory attached to the dermatology department as per the institutional policy. We had antenatal COVID-19 suspects and positive cases in our hospital, who did not have documented reports of VDRL test. We perform VDRL tests, taking all the precautions recommended by the World Health Organization (WHO).[4-6]
The specimens are well packed and labeled as biohazard.
Eating, drinking, smoking, applying cosmetics, and handling contact lens are prohibited in the laboratory working areas. Good housekeeping practices, sufficient space for specimen storage, and waste disposal are ensured.
As per the WHO guidelines, routine blood investigations should be performed in appropriately maintained and validated Class 2 biosafety cabinets.[4] BSL-2 protection is recommended by the CDC also. As per the present WHO guidelines on rational use of PPE, the laboratory technician needs to wear medical mask, goggles, gloves, and gown.[5] About 0.1% (1000 parts per million) hypochlorite is recommended to inactivate COVID-19.[4] Contact time recommended is 1–10 min.
For large blood spillage of >10 ml, a concentration of 0.5–1% hypochlorite is recommended.
All the wastes irrespective of the type of material are discarded in the yellow kit.
We do the VDRL tests following these precautions. The laboratory is informed about sending the sample, about 2–3 hours beforehand to provide sufficient time for taking the precautions. Health inspector is also informed about the test so that his team can be ready with the facilities to transport the waste materials after the procedure. The sample is triple packed and send to the laboratory. The laboratory technician wearing the PPE, receives it, and performs the test. The laboratory is closed during the entire procedure. Once the procedure is completed, a donned cleaning staff cleanses the laboratory with bleaching powder. All the wastes irrespective of the type are disposed in the yellow kit. This kit is then dumped in a yellow bag and taken away from the hospital premises in a vehicle immediately, by a team under the guidance of health inspector. After the procedure, we leave the laboratory closed for 2 days as COVID-19 virus may remain over inanimate surfaces for many hours. Fumigation is not done as it is found ineffective by the WHO.
Dermatological problems encountered in this pandemic during our COVID duties
Among health care workers
We had health care workers including doctors and staff nurses who presented with allergic reaction to some of the protective equipment used. The main culprits were sanitizers, detergents, goggles, N95 mask, gloves, and PPE. The clinical presentations ranged from mild pruritus to acute contact urticaria, angioedema, and detergent dermatitis. They were managed with emollients, antihistamines, topical steroids, and short course of steroids depending on the severity of the dermatitis.
Among COVID-19 patients
A 48-year-old patient, known case of psoriasis vulgaris, under remission for the past 6 months, presented with an acute exacerbation, 1 week after the successful treatment of COVID-19. He was managed symptomatically, but 5 days later, the condition progressed to erythrodermic psoriasis [Figure 1]. The possible triggering mechanisms for the exacerbation and the rapid deterioration could be the viral infection, mental stress, and hydroxychloroquine given for the management of COVID-19. The other dermatological problems encountered among COVID-19 patients were viral exanthematous rash, miliaria crystallina (probably precipitated by high-grade fever), and herpes zoster [probably precipitated by stress, Figure 2].
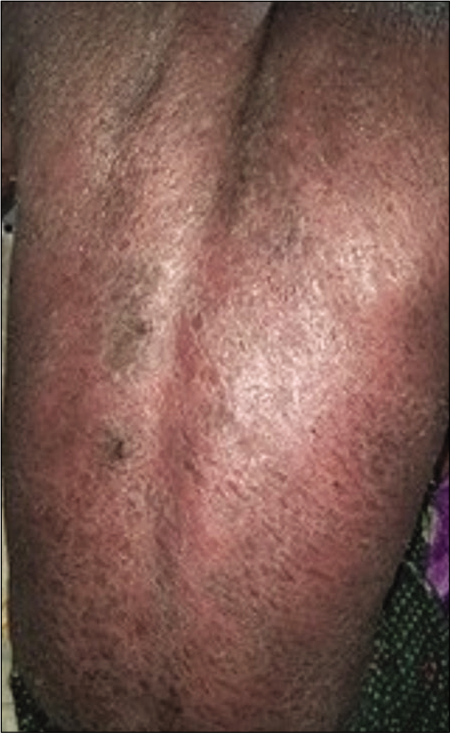
- Exacerbation of psoriasis to erythroderma in a patient after COVID-19 infection.
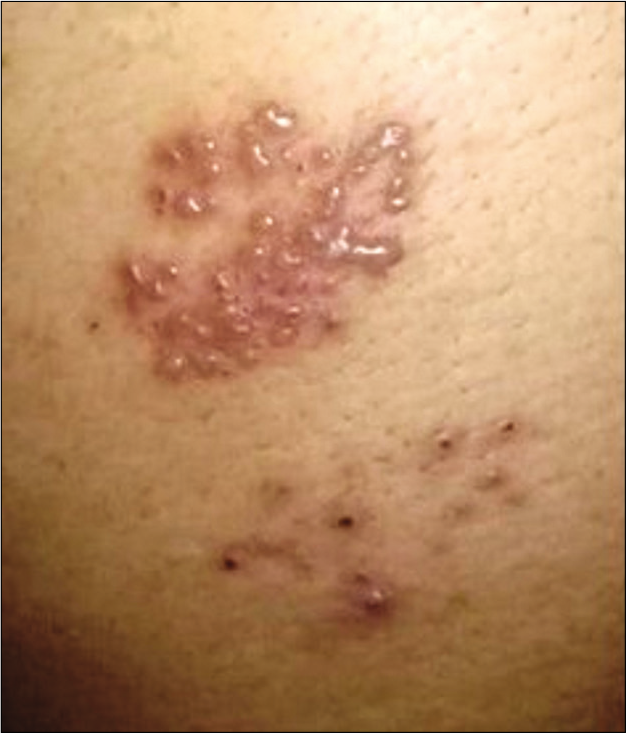
- Herpes zoster in a patient with COVID-19 infection.
CONCLUSION
The corona era is teaching us lessons of survival everyday. Recommendations are changing day by day. Timely interventions, anticipating the possible mishaps, are needed to tackle the collateral damages of COVID-19. We started the follow-up of vesiculobullous diseases on March 15, 2020. One week later, the statewide lockdown was declared, making it more difficult or even impossible for the patients to reach the hospital. Our strategies taught us volumes about the need for psychological support and patient care in this chaos of COVID-19. These measures also helped the patients to cope up with the difficult situation in a better way, by joining their hands with us in this battle against corona.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
Dr. Kunjumani Sobhanakumary is on the editorial board of the Journal.
References
- Impact of COVID-19 pandemic on dermatologists and dermatology practice. Indian Dermatol Online J. 2020;11:328-32.
- [CrossRef] [Google Scholar]
- Pemphigus. S2 guideline for diagnosis and treatment-guided by the European dermatology forum (EDF) in cooperation with the European academy of dermatology and venereology (EADV) J Eur Acad Dermatol Venereol. 2015;29:405-14.
- [CrossRef] [PubMed] [Google Scholar]
- Recommendations for phototherapy during the COVID-19 pandemic. J Am Acad Dermatol. 2020;83:287-8.
- [CrossRef] [PubMed] [Google Scholar]
- Laboratory Bio-Risk Management for Laboratories Handling Human Specimens Suspected or Confirmed to Contain Novel Coronavirus: WHO Interim Recommendations.
- [Google Scholar]
- Rational Use of Personal Protective Equipment for Corona Virus Disease (COVID-19) and Considerations during Severe Shortages. 2020. Geneva: World Health Organization; Available from: https://www.apps.who.int/iris/handle/10665/331695 [Last accessed on 2020 Apr 06]
- [Google Scholar]
- Laboratory Biosafety Guidance Related to Coronavirus Disease 2019 (COVID-19) Geneva: World Health Organization; 2020.
- [Google Scholar]






