Translate this page into:
Primary cutaneous CD30+ Anaplastic large cell lymphoma
*Corresponding author: Eapen Thomas, Department of Dermatology, Rajagiri Hospital, Chunangamvely, Aluva, Cochin, Kerala, India. eapenthomas10@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Thomas E, Abraham LK, Paul M. Primary cutaneous CD30+ Anaplastic large cell lymphoma. J Skin Sex Transm Dis 2020:2(2):144-5.
A 58-year-old housewife presented with indurated asymptomatic skin lesions on the right arm and posterior aspect of the left leg for 4 months. From a nearby hospital, she received treatment with topical steroids without much response. Clinical examination revealed multiple erythematous indurated plaque and nodule distributed over the right arm [Figure 1] and posterior aspect of the left leg in popliteal fossa [Figure 2]. A crusted plaque was seen on the right thigh. Skin biopsy showed large lymphoid cells with moderate cytoplasm and scattered mitotic figures [Figures 3 and 4]. Inflammatory infiltrates extended up to the subcutaneous tissue. Immunohistochemistry showed diffuse staining for CD30 [Figure 5] and focal staining for CD4. The inflammatory cells were negative for CD3, CD8, CD20, CD15, and ALK (anaplastic lymphoma kinase). Ki67 index was about 80%–90% in large cells. With a diagnosis of primary cutaneous CD30+ anaplastic large cell lymphoma, she was started on the CHOP regimen from the hematology department.
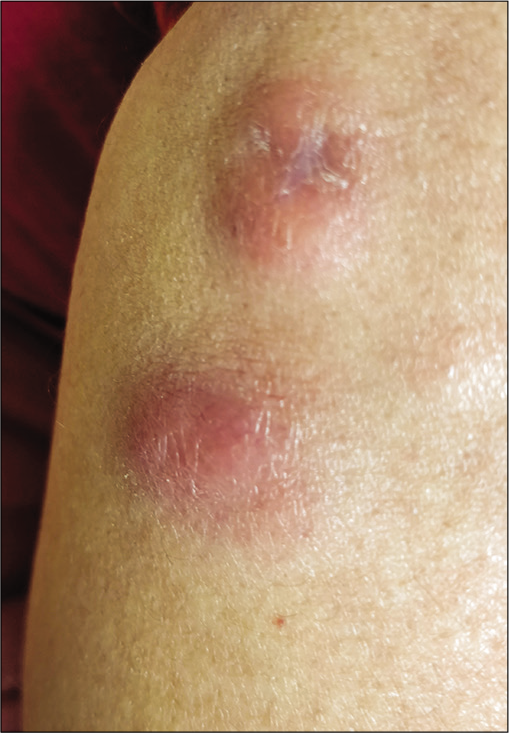
- Indurated nodule on the forearm.
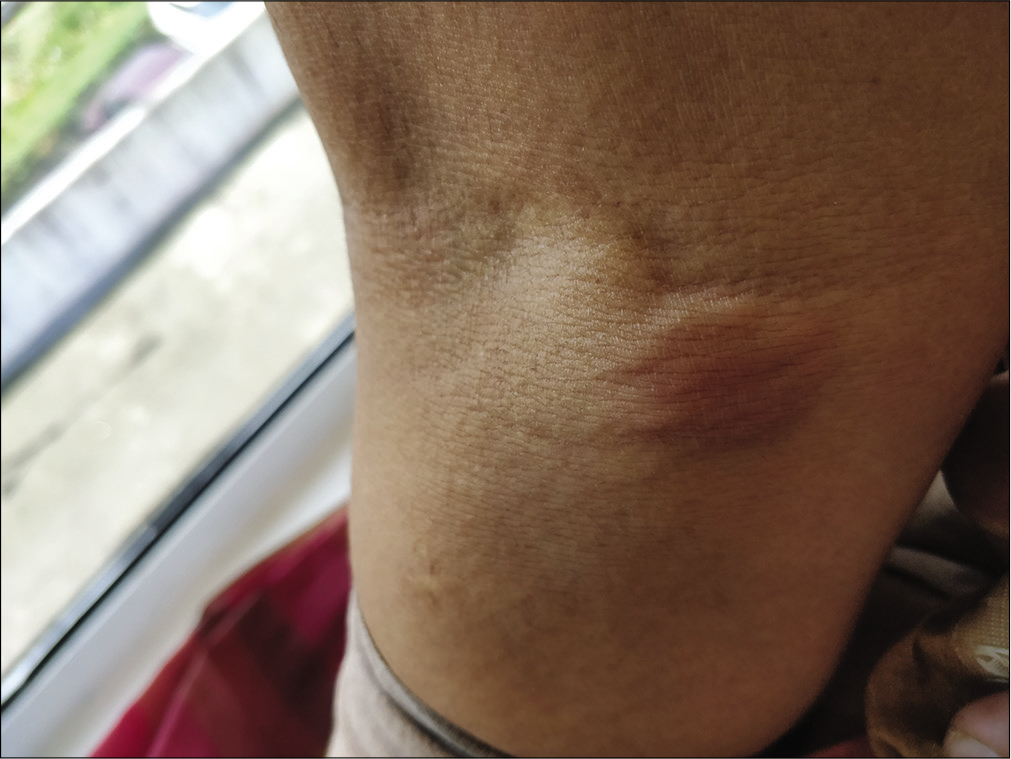
- Erythematous nodule on the posterior aspect of the leg.
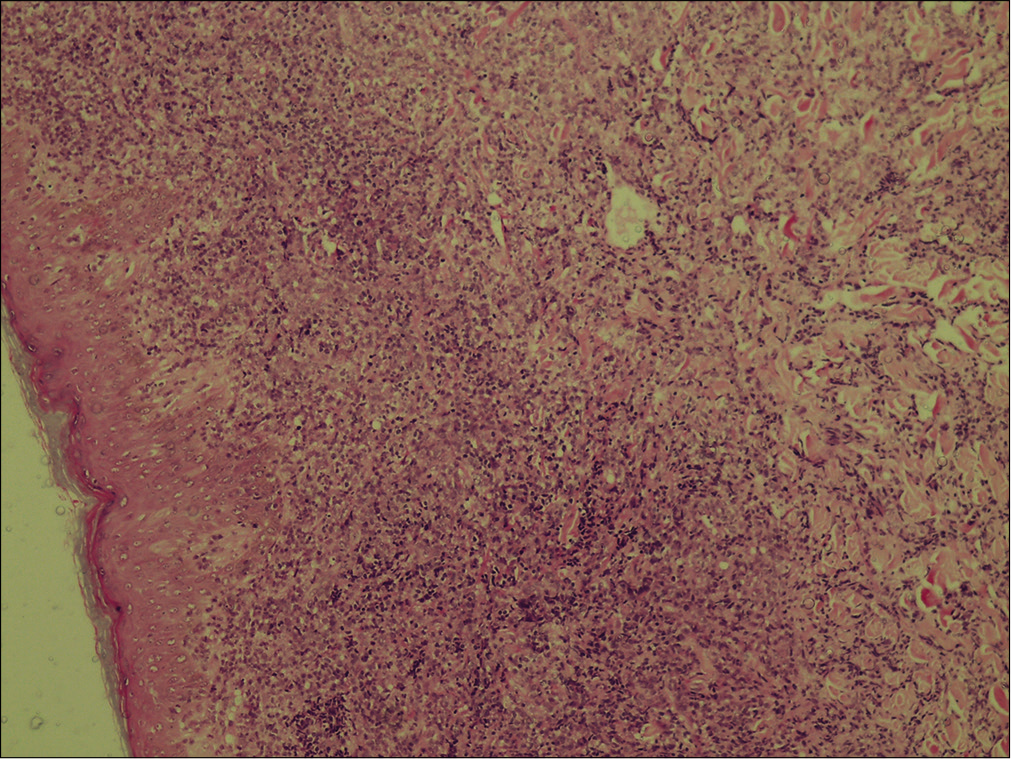
- Large lymphoid cells with moderate cytoplasm and scattered mitotic figures. Inflammatory infiltrate extending up to the subcutaneous tissue (H & E, ×100).
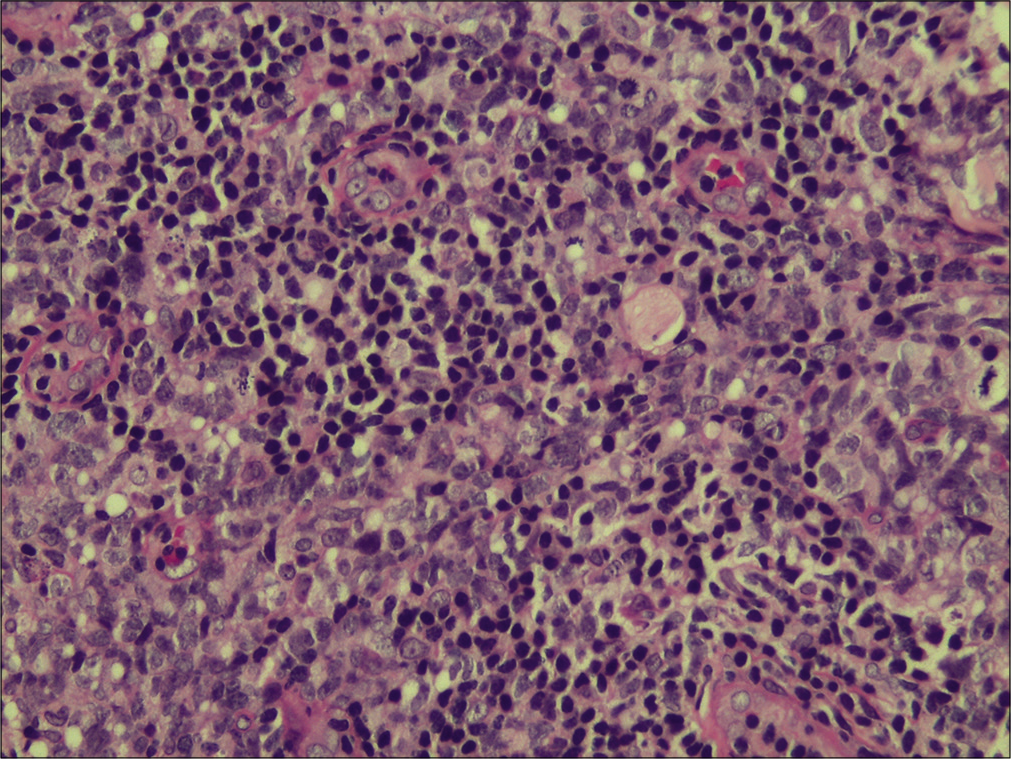
- Higher magnification showing the large lymphoid cells and the mitotic figures (H & E, ×400).
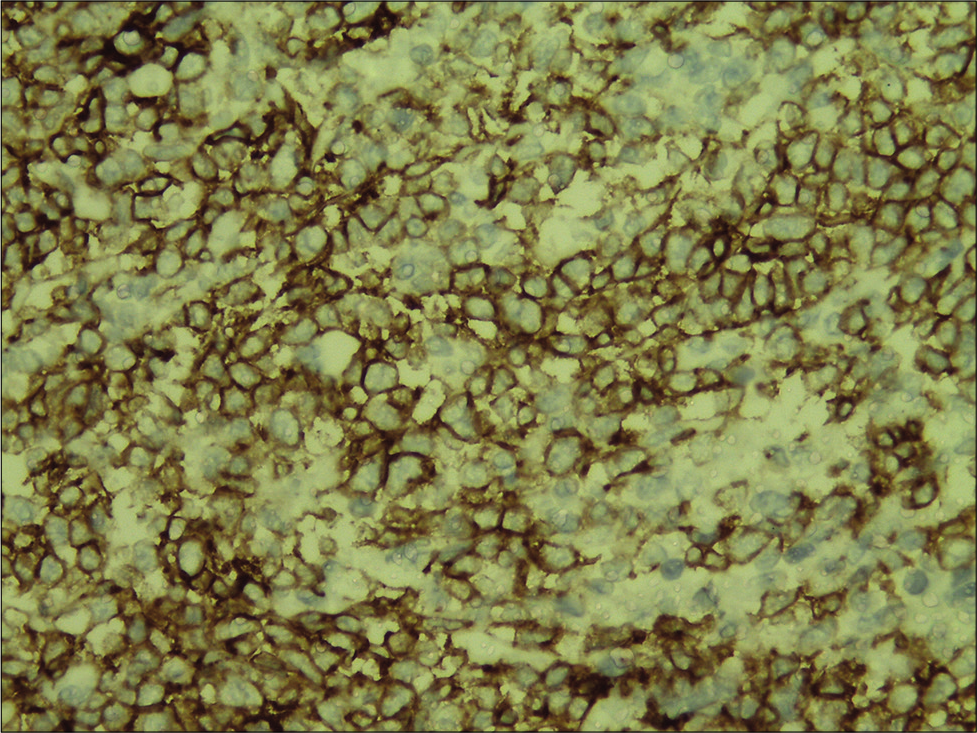
- Inflammatory infiltrate showing strong and diffuse staining for CD30 (immunohistochemistry, ×400).
Anaplastic large cell lymphoma (ALCL) represents a generally recognized group of large cell lymphomas. Defining features consist of a proliferation of predominantly large lymphoid cells with a strong expression of the cytokine receptor CD30 and a characteristic growth pattern. Cutaneous CD30 positive lymphoproliferative disorders may be primary (primary cutaneous ALCL) or secondary (systemic ALCL). Anaplastic large cell lymphoma can be CD30+ or CD30−. CD30 is a member of the cell surface receptors for tumor necrosis factor. The histologic clue to the diagnosis of ALCL, cutaneous or systemic, is the presence of large lymphoid cells, seen in the case presented and also known as “hallmark cells,” with embryonal nuclei, multiple prominent eosinophilic nucleoli, and abundant cytoplasm.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.





