Translate this page into:
Analysis of mortality and loss to follow-up during the rollout of the second-line antiretroviral therapy: An observational study from South India
*Corresponding author: Dr. P. C. Amrutha, Udaya, Nut Street, Vatakara, Kerala, India. amruthapc@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Ajithkumar K, Amrutha PC, Vinitha UC, Rakesh TP, Andrews AM. Analysis of mortality and loss to follow-up during the rollout of the second-line antiretroviral therapy: An observational study from South India. J Skin Sex Transm Dis 2019;1:19-23.
Abstract
Background
Assessment of the functioning of Kerala’s second-line component of antiretroviral therapy (ART) program by National AIDS Control Organization (NACO) is attempted.
Aims
This study aims to evaluate the factors related to mortality and loss to follow-up (LFU) during the second-line ART rollout in Kerala.
Materials and Methods
Prospective observational cohort study. All patients referred for the second-line ART from September 2011 to June 2013 were included, followed up till December 2013 or till death. Those who were not eligible to attend the State AIDS Clinical Expert Panel (SACEP) were excluded from the study. Descriptive variables were compared using proportions and percentages. Univariate analysis and multivariate logistic regression were done to find out the association. The study was approved by ethical committee and consent was taken from all the interviewed patients.
Results
Of 238 patients enrolled, 62 died and 25 became LFU. Age >40 years (odds ratio [OR] 2.08; 95% confidence interval [CI]: 1.05, 4.1), HIV concordance between partners (OR 1.53; 95% CI: 0.7, 3.34), and duration of >90 days from last CD4+ to SACEP (OR 2.32; 95% CI: 1.17, 4.5) were significantly associated with death. Only factor affecting LFU was distance >150 km from patient’s home to ART Plus Centre (OR: 2.7; 95% CI: 0.11, 1.85).
Limitations
We could not consider all factors affecting mortality while initiating second-line ART. Moreover, experience from a low-level epidemic state, with good health-care infrastructure may not reflect rest of India.
Conclusions
Accessibility to program in terms of distance to the point of care and delayed linkage of patients for the second-line ART is presently the main weaknesses in Kerala. Special attention should be given to concordant couples and older individuals who are more vulnerable. Recent steps by NACO, such as initiation of two ART Plus Centre and provision viral load testing at the point of care, are big leaps toward solution.
Keywords
Second-line antiretroviral therapy
Mortality
Loss to follow-up
India
INTRODUCTION
First decade of the 21st century saw rapid scale-up of antiretroviral therapy all over the world. Countries including India have rolled out antiretroviral therapy (ART) extensively sooner to be followed by issues of drug resistance and need for the second-line ART. In a country like India, designing and implementing a national public health program without compromising on individualized patient care is a challenge. Rising up to this, India established its second-line ART in 2008 which is a well-run and widely accepted program. However, it requires further evaluation regarding effectiveness and for improvisation. This study is a reflection on the second-line program in Kerala, which is functioning under the National AIDS Control Organization (NACO).
In the present model of national program being followed, due to the lack of uniform availability of viral load and drug sensitivity tests, NACO uses clinical evaluation, CD4+ cell count levels, and targeted viral load for identifying eligible patients for the second-line treatment. The responsibilities of the selection of patients eligible for the second-line ART and their management for the first 6 months are entrusted with referral centers, namely ART Plus Centres.
Kerala, a South Indian state, has a population of 3,404,050 and HIV prevalence of 0.1%.[1] In 2004, Kerala started its own free ART program which was later integrated with the national program. ART Plus Centre at our institution functions as referral center for the eight ART centers of the state and follows NACO guidelines for provision of the second-line ART. From all ART centers, patients who fulfill the criteria for clinical and immunological failure are referred here. A trained clinician here reviews the case records and selects the patients eligible for attending State AIDS Clinical Expert Panel (SACEP) constituted by a group of experts in HIV management, where adherence-related issues, opportunistic infections, and factors related to the first-line failure are evaluated. Those who are on the first-line ART for the past 6 months, showing immunological failure in spite of good adherence (evidenced by the development of new opportunistic infections or fall of CD4+ cell count to a level lower than baseline or half of the maximum achieved or failure to achieve a CD4+ cell count >100 cells/mm3 after a year of regular treatment) are referred for viral load. The second-line ART is initiated if viral load is >1000 copies per ml.[2] These patients are followed up at the center for the next 6 months. Viral load is repeated after 6 months of the second-line ART and patients who achieve viral suppression and are clinically stable referred back to their respective ART centers for continued care.
MATERIALS AND METHODS
Aim
The aim of this study was to evaluate factors related to mortality and loss to follow-up (LFU) during the rollout of the second-line ART program.
Objective
The objectives of this study were to assess the relationship of death and LFU to factors such as age, gender, CD4+ at failure detection, distance from patient’s home to ART Plus Centre, marital status, HIV concordance between partners and duration between last CD4+ and SACEP, and also finding out the retention in care. We defined retention in care as the percentage of patients who were alive on the second-line ART at the end of the study period, which is 6 months after enrollment the last patient.
All patients referred to attend SACEP from September 2011 to June 2013 for consideration of initiating second-line ART were included in this study and were followed up till December 2013 (6 months after the recruitment of the last patient) or till death. Those who were not eligible for SACEP due to lack of adherence were excluded from the study. The total follow-up period was 1603 person months, mean follow-up 13 months with a range from 6 months to 26 months. LFU is defined as not attending ART clinic for >3 consecutive months. Death was confirmed from records of ART centers and/or the information from immediate relatives of the patients. We looked for possible relationship of LFU and death with factors such as age, gender, CD4+ at failure detection, distance of ART Plus Centre from their home, marital status, HIV concordance between partners, and time gap between last CD4+ and SACEP. The study was approved by the ethical committee of our institution and consent was obtained from all the interviewed patients.
We collected the demographic data, details of first-line ART, marital status, adherence, previous CD4+ counts, distance from nodal ART center and HIV status of the spouse from referral letters, registers, reports of SACEP meetings, and records from the clinic and also from interview by a trained social scientist at the time of their clinic visit. Data regarding patients who are LFU at any stages of care were collected either from their nodal ART center or telephonically from patient or relatives. Patients received financial aid from Kerala State AIDS Control Society for travel through their parent ART centers. Data were analyzed using Excel© and PSPP version 0.10 software. Univariate analysis was done first and significant outcomes were analyzed using multivariate logistic regression. P < 0.05 was taken as statistically significant.
RESULTS
A total of 258 patients were referred to our center; among them, 238 were eligible referrals. The overall outcome, as in death and LFU at various stages are depicted in the flowchart [Chart 1]. Of the 238 patients, only 220 (92.4%) attended SACEP, main reason for non-attendance being death before that. After evaluation, viral load testing was done in 182 patients and 132 (55.4%) were eligible for the second-line ART. It was initiated in 123 patients (51.6%). Seven patients expired and two patients became LFU before second-line ART could be started. At the end of the study period, 102 patients were alive on second-line ART and the retention in care was found to be 42.8%.
Table 1 shows the demographic profile of the patients. Table 2 shows the results of univariate analysis and Table 3 displays the results from multivariate analysis. 62 of the 238 patients who were selected to attend SACEP died while in the process of evaluation and initiation of the second-line ART (27 before the initiation of second line, 17 after initiation of second line, and 18 deaths occurred among those who were excluded). 11.3% (27 of 238) of the eligible referrals died before initiation of the second line at various levels. Death is significantly associated with the age >40 years (odds ratio [OR] 2.08; 95% confidence interval [CI]: 1.05, 4.1), HIV concordance between partners (OR 1.53; 95% CI: 0.7, 3.34), and duration of >90 days between last CD4+ cell count measurement and SACEP attendance (OR 2.32; 95% CI: 1.17, 4.5). Only one factor was shown to be associated with LFU and it was distance of >150 km from home to ART Plus Centre (OR: 2.7; 95% CI: 0.11, 1.85). Of 123 patients initiated on the second-line ART, seven were referred for clinical failure, 18 for clinical and immunological failure, and 85 for immunological failure. 13 patients were initiated on the second line before referral by referring centers.
| Characteristics | Number of patients (%) |
|---|---|
| Gender | |
| Male | 178 (68.9) |
| Female | 80 (31.00) |
| Age (years) | |
| 0–20 | 11 (4.2) |
| 21–30 | 18 (6.9) |
| 31–40 | 91 (35.27) |
| 41–50 | 92 (35.65) |
| 51–60 | 34 (13.17) |
| 61–70 | 12 (4.6) |
| Distance (km) | |
| <50 | 60 (23.2) |
| 50–100 | 20 (7.7) |
| 100–150 | 101 (39.1) |
| 150–200 | 18 (6.9) |
| 200–300 | 54 (20.9) |
| >300 | 5 (1.9) |
| Feature | P value | ||
|---|---|---|---|
| Death | Loss to follow-up | Death after start in second-line ART | |
| Gender | 0.45 | 0.16 | 0.94 |
| Male | |||
| Female | |||
| Age (years) | 0.04 | 0.09 | 0.02 |
| >40 | |||
| <40 | |||
| CD4+ at failure detection | 0.56 | 0.88 | 0.51 |
| <200 | |||
| >200 | |||
| Distance (km) | 0.07 | 0.03 | 0.01 |
| >150 | |||
| <150 | |||
| Presence of spouse | 0.53 | 0.58 | 0.61 |
| Yes | |||
| No | |||
| HIV concordance | 0.04 | 0.8 | 0.29 |
| Yes | |||
| No | |||
| Duration between last CD4+ and SACEP | 0.01 | 0.8 | 0.14 |
| >90 days | |||
| <90 days | |||
| Feature | Death | Loss to follow-up | Death after starting second-line ART | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B | Exp B | 95% CI | P value | B | Exp B | 95% CI | P value | B | Exp B | 95% CI | P value | |
| Age | 0.732 | 2.08 | 1.05, 4.1 | 0.04 | 1.61 | 5 | 1.01, 47.31 | 0.02 | ||||
| >40 | ||||||||||||
| <40 | ||||||||||||
| Distance | −0.63 | 0.53 | 0.28, 1.01 | 0.054 | 1.01 | 2.7 | 0.11, 1.85 | 0.03 | 1.23 | 3.44 | 0.28, 20.16 | 0.03 |
| >150 km | ||||||||||||
| <150 km | ||||||||||||
| HIV concordance | 0.43 | 1.53 | 0.7, 3.34 | 0.01 | ||||||||
| Yes | ||||||||||||
| No | ||||||||||||
| Duration between last CD4+ and SACEP | 0.84 | 2.32 | 1.17, 4.5 | 0.02 | ||||||||
| >90 days | ||||||||||||
| <90 days | ||||||||||||
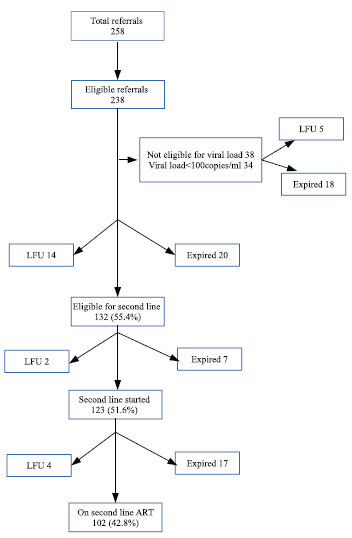
- Outcome of patients referred for second line anti retroviral therapy.
About 13% (17 of 123) of patients initiated on the second line died within the study period. Moreover, the factors affecting death after starting second-line ART were age >40 years (OR 5; 95% CI: 1.01, 47.31) and distance of >150 km from patient’s home to ART Plus Centre (OR 3.44; 95% CI: 0.28, 20.16).
DISCUSSION
In our study, among the 258 referrals for the second line, 20 did not qualify for SACEP evaluation (7.7%). This reiterates the need for a second level scrutiny of patients selected for the second-line ART. Non-adherence to drugs is a key issue in ART, leading to treatment failure, changing regimens without addressing this would be futile. In a study done in South Africa, the second-line ART failed to suppress the viral load in 39% of patients.[3] Adherence is also a predictor of future second-line failure.[4] Thus, just the universal availability of viral load test, though absolutely essential, does not become the panacea and it cannot replace good patient-doctor and patient-counselor interaction and adopting aggressive measures to address patient-related factors, leading to poor adherence. Viral load should preferably be done for patients with good adherence and this study underscores the need for reevaluation of patients selected for viral load test.
Our study showed the factors affecting mortality as age >40 years, HIV concordance among partners, and duration of >90 days between last CD4 and SACEP attendance. The relationship between age and adverse clinical outcome in HIV is already known.[5] Few international studies to highlight the relationship between age and adverse outcome.[6,7] Similarly, a relationship between the HIV concordance and bad outcome is already reported from this region.[8] The duration of >90 days between last CD4+ cell count measurement and SACEP can be taken as a surrogate of time taken for linkage, though there are multiple factors, even patient related, delaying the linkage. This delay in linkage, and hence, delay in switching of ART is an important contributor for future failure of the second-line therapy and will increase the mortality.[9] In another cohort of patients from Rwanda, mortality rate was high among those who were not switched and CD4 decline, and virological failure was high among those who were linked beyond 4 months.[10] Even small delays were associated with marked increase in mortality and future second-line failure as proposed by the marginal structural models in one study from South Africa.[11] There should be more studies to identify the factors delaying the linkage, both patient and provider related and interventions should be focused on.
Our study had death of 13% (17 of 123) after initiation of the second-line ART within the study period. There are some studies showing good outcomes of the second-line ART from India.[12] Increased death rate during the 1st year after initiation of the second-line ART has also been reported previously from the country. Similarly, some factors such as adherence and delayed initiation of the second line have also shown association with increased mortality during the second-line ART.[13] We could not assess all the factors affecting mortality as reliable data were not available, even though certain contributing factors such as delayed linkage and difficulties faced by concordant couples are seen in this study. Patients may also become too sick to travel. Establishment of two more ART Plus Centres in the state along with the provision of viral load facilities at ART centres are positive steps towards resolving this difficulty faced by patients.
From our study, the only factor affecting LFU was found as the distance of >150 km from home to ART Plus Centre. Among them, only 6.7% (16 of 238) of patients became LFU after expert panel selection for viral load testing. This possibly implies those who can reach expert panel can also reach viral load facility and initiate and sustain second-line ART even though viral load testing is >500 km away. Those who are less empowered may have already been eliminated from the flow at the level of referral to the ART Plus Centre itself. This aspect needs to be looked into in detail to take adequate measures to reduce drop out from the system. This literature review clearly depicts the role of transportation difficulties in pre-ART care and in linkage of patients to ART.[14] Ironically, the situation repeats itself even at the advanced stages of delivery of care. The steps taken by NACO to make point of care viral load test will solve this problem to a great extent. After initiation of the second-line ART, there were only four LFUs, which are 3.2% of the total number initiated. This is a commendable achievement as it would help reduce the failure rate and need for an expensive third-line therapy.
This study is undertaken with the perspective of effectiveness of the national program and hence limited in the aspect of analysis of all factors affecting mortality while initiating patients on second-line ART. The associations shown here are multifactorial, contributed by programmatic as well as patient related factors. Furthermore, experience from a state with low-level epidemic, few patients and a rather well-run first-line ART program in the background of comparatively good health-care infrastructure may not be represent the reality of the rest of India.
At present, there are limited studies from India on the second-line component of national ART program and the need for the same is paramount in the wake of India providing third-line ART. This study has looked into the strengths and weaknesses of the second-line rollout in Kerala state. It has brought forth certain issues such as increased death rate, delayed linkage, and the consequences of lack of widespread availability of facilities. Our conclusion is that accessibility to the program in terms of distance to the point of care and delayed linkage of patients for the second line is the main weakness in the current second-line program of Kerala. Furthermore, there are older patients and concordant couples whose problems need to be addressed to reduce mortality. Various national and international studies point out similar factors such as adherence and limitations of a centralized mode of delivery of care as the main causes of failure even during the second-line therapy.[3,15] Goal-directed approach in these areas like enhanced adherence support has been found to be fruitful.[16] Many programmatic aspects are being taken care of by NACO. The essence is the need for scale-up of the program in respect to more referral centers and viral load facilities without compromising patient-provider interaction.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- HIV in Kerala In: Rajan SI, Zachariah KC, eds. Kerala’s Demographic Future Issues and Policy Options. Thiruvananthapuram: Centre for Development Studies; 2012. p. :387-96. In: p.
- [Google Scholar]
- Second Line Anti Retroviral Therapy Related Activities Operational Guidelines for Anti Retroviral Therapy Services. 2013. New Delhi: National AIDS Control Organization; Available from: [Last accessed on ] (accessed )http://www.naco.gov.in/care-support-treatment
- [Google Scholar]
- Social and behavioral factors associated with failing second-line ART-results from a cohort study at the themba lethu clinic, Johannesburg, South Africa. AIDS Care. 2018;30:863-70.
- [CrossRef] [PubMed] [Google Scholar]
- Predicting the need for third-line antiretroviral therapy by identifying patients at high risk for failing second-line antiretroviral therapy in South Africa. AIDS Patient Care STDS. 2017;31:205-12.
- [CrossRef] [PubMed] [Google Scholar]
- Determinants of survival in adult HIV patients on antiretroviral therapy in eastern Uttar Pradesh: A prospective study. Ind J Med Res. 2014;140:491-500.
- [Google Scholar]
- Survival and causes of death among HIV-infected patients starting antiretroviral therapy in North-Eastern Vietnam. Scand J Infect Dis. 2012;44:201-8.
- [CrossRef] [PubMed] [Google Scholar]
- Determinants of mortality and loss to follow-up among adults enrolled in HIV care services in Rwanda. PLoS One. 2014;15:e85774
- [CrossRef] [Google Scholar]
- Relationship between social factors and treatment adherence: A study from South India. East J Med. 2011;16:147-52.
- [Google Scholar]
- Delayed switch of antiretroviral therapy after virologic failure associated with elevated mortality among HIV-infected adults in Africa. AIDS. 2014;28:2097-107.
- [CrossRef] [PubMed] [Google Scholar]
- Rates of switching to second-line antiretroviral therapy and impact of delayed switching on immunologic, virologic, and mortality outcomes among HIV-infected adults with virologic failure in Rakai, Uganda. BMC Infect Dis. 2017;17:582.
- [CrossRef] [PubMed] [Google Scholar]
- Marginal structural models to assess delays in second-line HIV treatment initiation in South Africa. PLoS One. 2016;11:e0161469.
- [CrossRef] [Google Scholar]
- An early outcome of second line antiretroviral therapy in treatment-experienced human immunodeficiency virus positive patients. Perspect Clin Res. 2013;4:215-20.
- [CrossRef] [PubMed] [Google Scholar]
- Outcome of patients on second line antiretroviral therapy under programmatic condition in India. BMC Infect Dis. 2015;15:517.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors, barriers and facilitators for linkage to antiretroviral therapy care. AIDS. 2012;26:2059-67.
- [CrossRef] [PubMed] [Google Scholar]
- Switching to second-line ART in relation to mortality in a large tanzanian HIV cohort. J Antimicrob Chemother. 2017;72:2060-8.
- [CrossRef] [PubMed] [Google Scholar]
- Second-line failure and first experience with third-line antiretroviral therapy in Mumbai, India. Glob Health Action. 2014;7:24861.
- [CrossRef] [PubMed] [Google Scholar]






